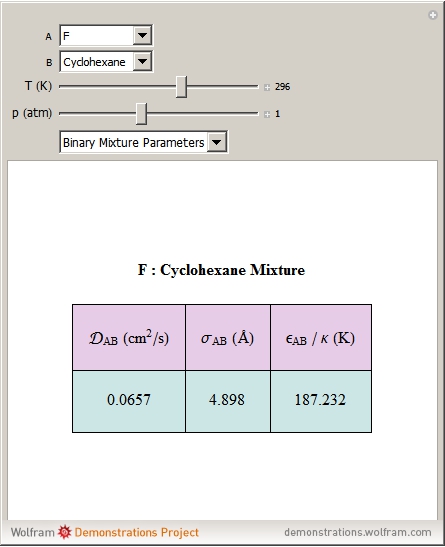
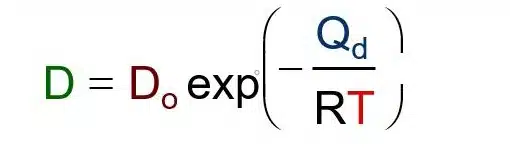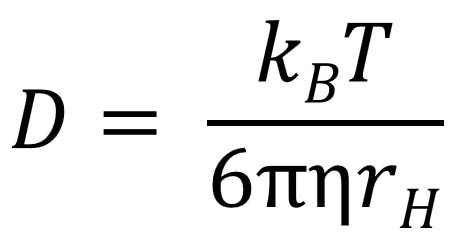SOLVED: According to the Kinetic Theory of gases, the diffusion coefficient; D, of a gas can be described using the following equation: 1 D = v^ 3 Using this ratio calculates the

Simple and accurate correlations for diffusion coefficients of solutes in liquids and supercritical fluids over wide ranges of temperature and density - ScienceDirect

Accounting for frame of reference and thermodynamic non-idealities when calculating salt diffusion coefficients in ion exchange membranes - ScienceDirect

The diffusion coefficient of an ideal gas is proportional to its mean free path and mean speed. The absolute temperature of an ideal gas is increased 4 times and its pressure is




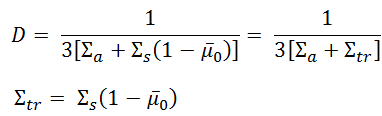

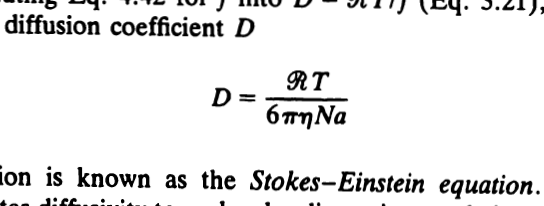

![Example: In the formula, N=−D[x2−x1n2−n1],D is called diffusion co.. Example: In the formula, N=−D[x2−x1n2−n1],D is called diffusion co..](https://static-images.findfilo.com/classroom/1678080968687_pvxacumf_1419100.jpg)