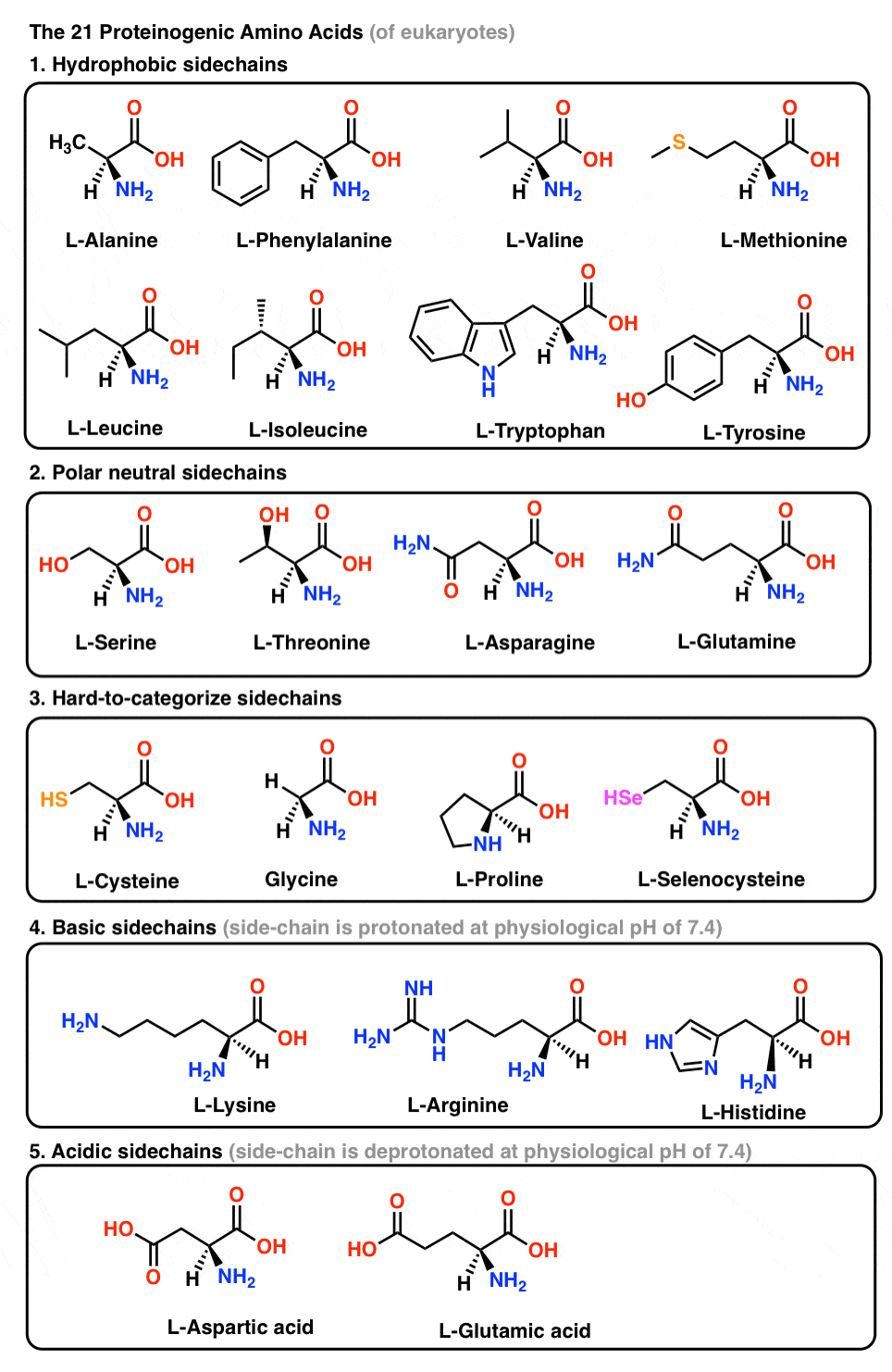
Molecules | Free Full-Text | Efficient Fmoc-Protected Amino Ester Hydrolysis Using Green Calcium(II) Iodide as a Protective Agent

Development of Fmoc-Protected Bis-Amino Acids toward Automated Synthesis of Highly Functionalized Spiroligomers | Organic Letters

An Fmoc protecting group can be removed from an amino acid by treatment with the amine base piperidine. Propose a mechanism. | Homework.Study.com
Fmoc-L-Proline, 50 g, CAS No. 71989-31-6 | Fluorenylmethylene / Fmoc | Amino acids, protected | Amino Acid Derivatives | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International

Fmoc-OASUD: A new reagent for the preparation of Fmoc-amino acids free from impurities resulting from Lossen rearrangement - ScienceDirect

Synthesis of Fmoc-protected 4-N,N,-dimethylaminophthalimidoalanine (1)... | Download Scientific Diagram
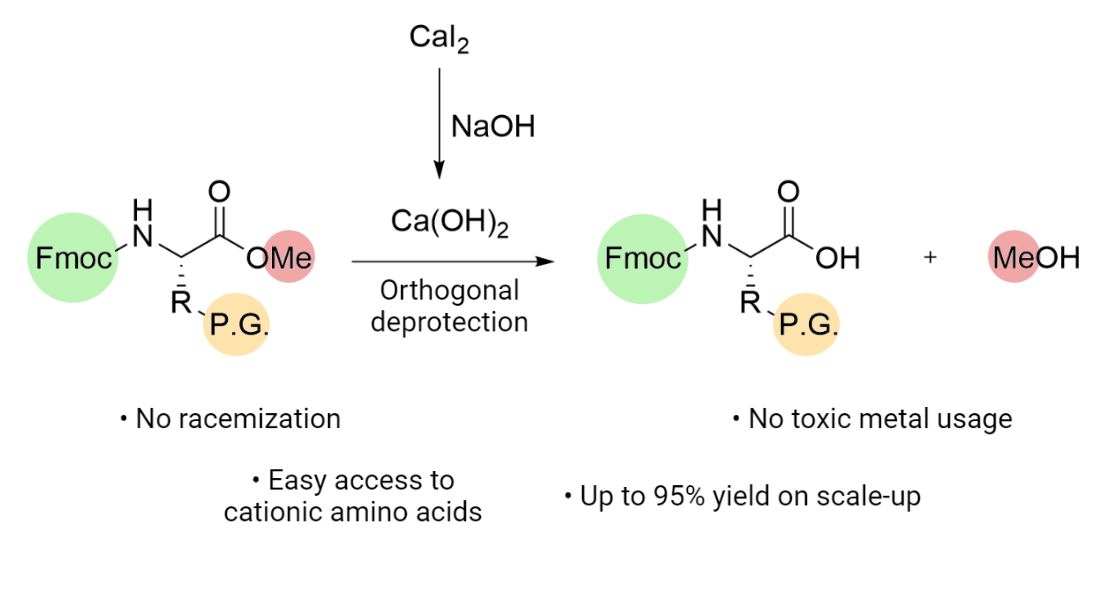
Molecules | Free Full-Text | Efficient Fmoc-Protected Amino Ester Hydrolysis Using Green Calcium(II) Iodide as a Protective Agent
Fmoc-L-Lysine-(Boc), 5 g, CAS No. 71989-26-9 | Fluorenylmethylene / Fmoc | Amino acids, protected | Amino Acid Derivatives | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International

Large-Scale Syntheses of FMOC-Protected Non-Proteogenic Amino Acids: Useful Building Blocks for Combinatorial Libraries | Organic Process Research & Development



![Fmoc-Amino Acids [N-Protected Amino Acids] | TCI AMERICA Fmoc-Amino Acids [N-Protected Amino Acids] | TCI AMERICA](https://www.tcichemicals.com/medias/B3669.jpg?context=bWFzdGVyfHJvb3R8MTYwMzl8aW1hZ2UvanBlZ3xoMmMvaDk0LzkwODY4NzA5Nzg1OTAvQjM2NjkuanBnfDZjMWQ5ZWRhMzRjODI0Y2JhNmY4MGE0MDFmOTM1NGZiZDk2ZmYxZGYzNzE0MDEyN2JlMWQ5MzI1NzU1ODg0ZDk)